
The big (and I mean BIG) astronomy news of the day is the announcement of possible (and I mean possible) signs of life on Venus—specifically, the detection of the molecule phosphine (PH3) in the upper atmosphere. The researchers, a large collaboration, put out two papers about it today: the original Nature Astronomy paper and a 103-page supplement explaining why all the alternative explanations that they could think of don’t work.
Predictably, the media are running with it, but does it mean there is life on Venus? As cool as that would be, right now, I would bet against it.
The upshot: it is very possible, even probable, that phosphine on Venus could have come from some non-life chemical process we haven’t figured out yet. It’s also possible that it was misidentified sulfur dioxide, because their spectra look similar.
So, what’s going on here?
Venus is well known as the hottest planet in the solar system, victim of a runaway greenhouse effect that renders its surface as hot as a pizza oven. However, there are two things that have caused people to speculate as far back as the 1960s that Venus could have life floating in the clouds. First, when Venus was young, the Sun was cooler, and the runaway greenhouse hadn’t started yet. Back then, we’re pretty sure Venus had oceans much like our own where life could have formed.
Second, even today, there is a region of Venus’s atmosphere that is still habitable. Venus is also known for having an atmospheric pressure that’s like being at the bottom of the ocean, but that means the atmospheric pressure is the same as Earth’s surface at a height of about 50 km (30 miles). And the temperature at this altitude is also habitable. Well, we wouldn’t care for it that much, but it’s below the boiling point of water. And there’s no oxygen, but plenty of bacteria thrive without oxygen. And it’s very acidic. And dry. But it’s still nothing some Earth life couldn’t handle.
On Earth, bacteria can survive blowing on the wind over 10,000 meters (33,000) feet high. So if Venus once did have life in its oceans, some of that life could have made it to the upper atmosphere and survived to this day. Basically, even though I’m betting against it, it wouldn’t surprise me much if there is life there.
Now, what is phosphine? Phosphine is PH3, a molecule chemically similar to ammonia* that is produced by some microorganisms. In fact, on Earth, it’s produced only by microorganisms. If we see a rocky planet like Earth that has phosphine in its atmosphere, it’s a good bet that there’s life on it, as Clara Sousa-Silva pointed out last year.
But Venus is not the kind of planet Sousa-Silva was talking about. Yes, there could be life surviving in the upper atmosphere, but with extraordinary claims, we should already be considering alternatives; and with a planet so different from Earth, we should especially consider alternatives. After all, phosphine does occur on Jupiter, produced by high heat in its depths of over 800 kelvins (1000 degrees Fahrenheit)—not much hotter than Venus.
This very question was asked during the press conference, and Drs. Sara Seager and William Bains said they are very sure that the processes happening on Jupiter are not happening on Venus, because you need lots and lots of hydrogen for them. And to their credit, they were also cautious about the whole paper and said they haven’t definitively discovered life because we can’t be completely sure without going there.
But I believe even making the claims they have, they’re overselling it, and there are two main reasons why I believe this. First there has been very little laboratory work on phosphine. Here’s a figure from the Nature Astronomy paper showing all the chemical reactions they modeled to figure out what the non-life production of phosphine should be.
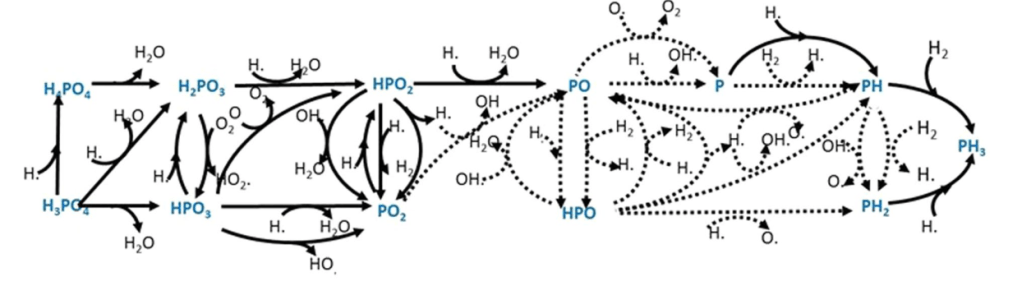
All those dotted lines are reactions that haven’t been done in a lab. The model is based on a worrying amount of guesswork, and we need more lab experiments to be confident about what’s going on.
Second, the group detected phosphine by its signature in the spectrum of light from Venus, but that signature overlaps with that of sulfur dioxide (SO2), which we do expect to find. The overlap isn’t perfect, and the authors say they ruled it out, but the thing is, they didn’t detect much SO2 at all when they probably should have. Venus is famous for its sulfuric acid clouds, and those are formed by reacting 150 ppm of SO2 with trace amounts of hydrogen chloride. The fact that it didn’t show up very well in the spectrum suggests that something strange is going on, and I consider it another point where there could be a mistake.
*But contrary to news reports, pure phosphine is odorless, unlike ammonia. Contamination with diphosphane (P2H4) gives it an unpleasant “garlic” or “rotting fish” odor.

Just posted on the same thing Alex, without your expertise and detail. Exciting though isn’t it?